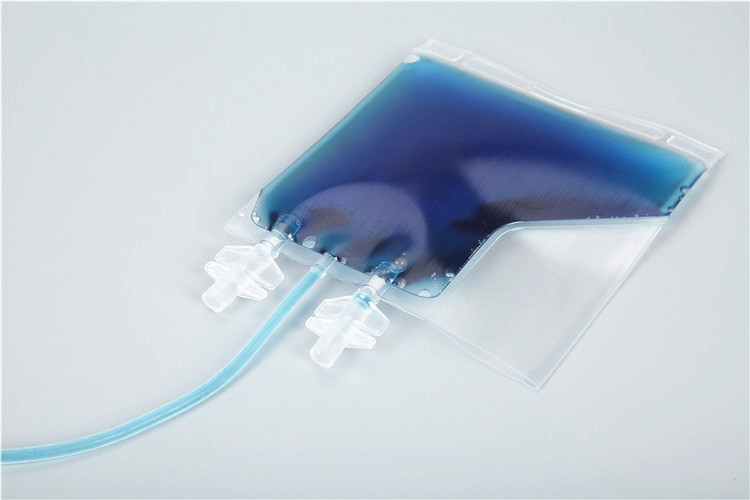
1. Overview of Cryopreservation Bags and Their Core Application Value
Cryopreservation bags are specialized flexible storage containers designed for the low-temperature preservation of biological samples, which can maintain the structural and functional integrity of samples under ultra-low temperature conditions (typically -80℃ to -196℃, liquid nitrogen environment). Different from conventional storage containers, cryopreservation bags must meet the dual requirements of biological safety and low-temperature stability, and can adapt to the harsh conditions of rapid freezing, long-term ultra-low temperature storage, and safe transportation, while avoiding sample contamination, leakage, or activity loss during the full storage process.
The core application value of cryopreservation bags lies in providing a closed, sterile, and low-damage storage solution for biological samples, which is closely linked to the entire industrial chain of biopharmaceuticals and clinical research. In the production of monoclonal antibodies, vaccines, and cell and gene therapies, cryopreservation bags are used to store seed cells, cell banks, and intermediate products; in clinical laboratories, they are used to preserve blood samples, tissue specimens, and stem cells; in scientific research, they are used to store experimental cells, microbial strains, and other biological materials. With the rapid development of cell and gene therapy technologies, the demand for cryopreservation bags with high performance, high safety, and traceability is increasingly prominent, and they have become an indispensable core auxiliary component in the field of biological sample management.
Compared with traditional storage methods (such as cryovials and stainless steel tanks), cryopreservation bags have obvious advantages: first, the flexible structure reduces the damage to samples caused by volume expansion during freezing (ice crystal formation); second, the large capacity (ranging from 1 mL to 5000 mL) adapts to the storage needs of different batch sizes of samples; third, the closed sterile design avoids cross-contamination between samples and the external environment; fourth, the lightweight and portable characteristics facilitate the transportation and transfer of samples; fifth, the compatibility with automated filling and storage systems improves the efficiency of sample management and reduces human error. These advantages make cryopreservation bags the preferred carrier for low-temperature storage of biological samples in modern biopharmaceutical and clinical fields.
2. Core Technology System and Material Characteristics of Cryopreservation Bags
2.1 Core Technical Principles
The core technical principle of cryopreservation bags is to realize the safe storage of biological samples by optimizing material selection, structural design, and sterile processing, and to minimize sample damage caused by low-temperature stress (ice crystal formation, osmotic pressure changes, and membrane damage). The realization of this principle relies on three key technical supports: low-temperature resistant material technology, closed sterile structural design, and cryoprotective compatibility technology, which together form the core technical system of cryopreservation bags.
First, low-temperature resistant material technology is the foundation of low-temperature storage. The materials of cryopreservation bags must maintain good flexibility and toughness under ultra-low temperature conditions, without brittleness, cracking, or leakage, and at the same time have excellent biocompatibility to avoid adverse reactions with biological samples. The material formula is usually modified by multi-layer composite technology to balance low-temperature resistance, biocompatibility, and mechanical strength. For example, the CryoStore® series cryopreservation bags adopt a multi-layer composite film structure, which can maintain structural integrity even after long-term storage in liquid nitrogen (-196℃) and avoid sample leakage.
Second, closed sterile structural design is the key to ensuring sample safety. Cryopreservation bags adopt a fully closed structure, with a sealed port (such as heat-seal port, screw port) that can effectively isolate the sample from the external environment, preventing microbial contamination and sample leakage during storage and transportation. The structural design also needs to consider the convenience of sample filling and sampling, and is usually equipped with a built-in sampling port or connection port, which can realize sterile sampling without opening the bag, further ensuring sample sterility. In addition, the surface of the bag is usually designed with a frosted area for marking sample information (batch number, storage time, sample type), facilitating sample traceability.
Third, cryoprotective compatibility technology is the guarantee of sample activity. Cryopreservation bags need to be compatible with common cryoprotectants (such as DMSO, glycerol, and serum), without chemical reaction with cryoprotectants or leaching harmful substances, so as to avoid affecting the activity of biological samples. The material selection and surface treatment technology of the bag are optimized to reduce the adsorption of cryoprotectants and sample components on the inner surface of the bag, ensuring the stability of the sample during long-term storage.
2.2 Key Material Characteristics and Classification
The material of cryopreservation bags directly determines their low-temperature resistance, biocompatibility, and safety, and is the core factor affecting sample storage quality. At present, the materials used in mainstream cryopreservation bags are mainly medical-grade multi-layer composite films, which are composed of base layer, barrier layer, and inner layer, each of which undertakes different functions:
- Inner layer: Directly in contact with biological samples, it must have excellent biocompatibility, non-toxicity, and no adsorption, and be compatible with cryoprotectants. Common materials include medical-grade polyethylene (PE), polypropylene (PP), and ethylene-vinyl acetate copolymer (EVA). Among them, EVA is widely used due to its good flexibility and low-temperature resistance, and can reduce the damage to samples caused by ice crystal formation during freezing.
- Barrier layer: Responsible for isolating oxygen, moisture, and other external substances, preventing oxidation and moisture absorption of samples during long-term storage, and maintaining sample activity. Common materials include polyvinylidene chloride (PVDC), polyethylene terephthalate (PET), and aluminum foil. The aluminum foil barrier layer has the best barrier effect, which is suitable for long-term storage of high-value samples (such as stem cells and gene therapy products).
- Base layer: Provides mechanical strength and wear resistance for cryopreservation bags, preventing the bags from being damaged during filling, storage, and transportation. Common materials include polyester (PET) and nylon (PA), which have good tensile strength and tear resistance, and can adapt to the harsh conditions of ultra-low temperature storage.
According to the material structure and application scenarios, cryopreservation bags can be divided into two main categories: single-layer cryopreservation bags and multi-layer composite cryopreservation bags. Single-layer cryopreservation bags are mainly made of PE or PP, with simple structure and low cost, suitable for short-term storage of non-critical samples (such as experimental cells). Multi-layer composite cryopreservation bags are made of 2-5 layers of composite films, which integrate the advantages of low-temperature resistance, barrier property, and biocompatibility, suitable for long-term ultra-low temperature storage of critical samples (such as cell banks, vaccines, and clinical samples). In addition, according to the storage temperature, they can be divided into -80℃ cryopreservation bags (for refrigerated storage) and -196℃ cryopreservation bags (for liquid nitrogen storage), which are selected according to the sample storage requirements.
2.3 Key Performance Indicators of Cryopreservation Bags
The performance of cryopreservation bags directly determines the quality and safety of biological sample storage. The key performance indicators recognized in the industry include the following aspects, which must meet the requirements of international standards (such as ISO 11607, ISO 10993) and global regulatory authorities (FDA, EMA, NMPA):
- Low-Temperature Resistance: It is the core indicator of cryopreservation bags, requiring that the bag body does not crack, brittle, or leak under the specified ultra-low temperature conditions (usually -80℃ to -196℃) after long-term storage (up to 5 years) and repeated freezing-thawing cycles (at least 5 times). The low-temperature resistance is verified by ultra-low temperature storage test and freezing-thawing cycle test to ensure the structural stability of the bag body under harsh conditions. For example, the CryoSure® cryopreservation bag can maintain structural integrity after 5 years of storage in liquid nitrogen (-196℃) and 10 times of freezing-thawing cycles.
- Biocompatibility: All materials in contact with biological samples must comply with the requirements of ISO 10993 series standards, without cytotoxicity, sensitization, pyrogenicity, hemolysis, and other adverse reactions, to avoid affecting the activity and quality of samples. Common biocompatibility tests include cytotoxicity test (ISO 10993-5), sensitization test (ISO 10993-10), pyrogen test (ISO 10993-11), and hemolysis test (ISO 10993-4), which must all pass the standard requirements. In addition, the materials must be free of animal-derived components (ADCF) and BPA, meeting the safety requirements of biopharmaceutical production.
- Sterility Assurance Level (SAL): Cryopreservation bags must be pre-sterilized before leaving the factory, and the sterility assurance level must reach ≤10⁻⁶, which is consistent with the sterility requirements of biopharmaceutical products. Common sterilization methods include gamma ray irradiation (25-50 kGy), ethylene oxide (EO) sterilization, and hydrogen peroxide plasma sterilization, among which gamma ray irradiation is the most widely used due to its high sterilization efficiency, uniform sterilization, and no residue. The sterilization process must be strictly controlled and verified to ensure that the sterility of the bag body is maintained before use.
- Sealing Performance: Under the conditions of ultra-low temperature storage, transportation, and freezing-thawing cycles, there is no leakage of the bag body and the sealed port. The sealing performance is verified by pressure holding test, vacuum test, and ultra-low temperature leakage test. For example, after filling the bag with simulated solution, it is stored in liquid nitrogen for 72 hours, and there is no leakage or seepage, which meets the sealing requirement. The heat-seal port of high-quality cryopreservation bags adopts hot-melt sealing technology, with uniform sealing strength and no edge leakage.
- Mechanical Strength: It has sufficient tensile strength, tear strength, and puncture resistance to avoid damage during filling, sealing, storage, and transportation. The tensile strength is usually required to be ≥20 MPa, and the tear strength ≥5 kN/m, which can withstand the external force impact during sample transfer and transportation. In addition, the bag body should have good flexibility, and no cracking or breaking when bent under ultra-low temperature conditions.
- Traceability: The surface of the bag body should be able to be marked with sample information (such as batch number, storage time, sample type) clearly and permanently, and the manufacturer should provide complete traceability documents, including material batch number, sterilization batch number, production date, and expiration date, to realize full-process traceability of samples. Some high-end cryopreservation bags are also equipped with RFID tags, which can realize intelligent management and traceability of samples.
- Cryoprotectant Compatibility: It is compatible with common cryoprotectants (such as DMSO, glycerol, serum, and trehalose), without chemical reaction with cryoprotectants, no leaching of harmful substances, and no adsorption of cryoprotectants, ensuring that the cryoprotective effect is not affected. Compatibility tests are usually carried out by soaking the bag body in cryoprotectant solution at ultra-low temperature for a long time, and detecting the changes of the solution components and the performance of the bag body.
3. Full-Process Application Guide of Cryopreservation Bags
The safe and effective application of cryopreservation bags involves the entire process from sample preparation, freezing, storage, transportation to thawing. Each link must be strictly operated in accordance with the standard procedures to ensure the quality and activity of biological samples. The following elaborates on the key operation points and precautions of each link to provide a practical application guide for users.
3.1 Sample Preparation and Bag Filling
Sample preparation and bag filling are the first links of cryopreservation, which directly affect the subsequent storage effect of samples. The key operation points are as follows:
- Sample Pretreatment: Before filling, the biological sample should be pretreated according to the sample type, including centrifugation, purification, and cryoprotectant addition. The cryoprotectant should be added slowly and evenly to avoid osmotic pressure shock to the sample; after adding, the sample should be incubated at 4℃ for 10-30 minutes to ensure that the cryoprotectant is fully mixed with the sample and penetrates into the cells, reducing ice crystal damage during freezing. For example, when storing stem cells, DMSO (final concentration 10%) is usually used as a cryoprotectant, which is added dropwise to the cell suspension and incubated at 4℃ for 20 minutes.
- Bag Selection and Inspection: Select the appropriate cryopreservation bag according to the sample volume, storage temperature, and storage time. Before filling, check the bag body for damage, leakage, or contamination, check the integrity of the sealed port, and confirm the sterilization batch number and expiration date of the bag body to avoid using unqualified products. For critical samples, it is recommended to use double-layer cryopreservation bags (inner bag for sample storage, outer bag for protection) to further reduce the risk of leakage.
- Sterile Filling: The filling operation should be carried out in a sterile environment (such as a biological safety cabinet) to avoid sample contamination. The filling volume should be controlled at 70%-80% of the bag capacity, leaving a certain space for volume expansion during sample freezing (the volume expansion rate of water during freezing is about 9%), preventing the bag body from bursting due to excessive pressure. After filling, the air in the bag should be exhausted as much as possible (avoiding vacuum to prevent the bag body from collapsing) to reduce the damage to the sample caused by air bubbles during freezing.
- Sealing and Marking: After filling and exhausting, the bag body should be sealed immediately. The sealing method should be selected according to the bag type: heat-seal bags should be sealed by a professional heat-sealing machine, controlling the sealing temperature (120-150℃) and time (2-5 seconds) to ensure uniform sealing strength; screw-port bags should be tightened to ensure no leakage. After sealing, mark the sample information (batch number, sample type, storage time, operator) on the frosted area of the bag body with a special marker (waterproof and low-temperature resistant), ensuring that the information is clear and not easy to fall off.
3.2 Sample Freezing Process
The freezing process is a key link to avoid sample damage. The core is to control the freezing rate to reduce the formation of large ice crystals (which will damage the cell membrane and tissue structure). The freezing rate should be adjusted according to the sample type, and the following key points should be noted:
- Freezing Rate Control: Different biological samples have different requirements for freezing rate. For example, the optimal freezing rate of mammalian cells is 1℃/min; the freezing rate of vaccines and biological reagents can be appropriately faster (2-5℃/min); the freezing rate of tissues and organs should be slower (0.1-0.5℃/min). The freezing rate can be controlled by a professional programmable freezer, which can realize the step-by-step cooling of samples from room temperature to -80℃, and then transfer to liquid nitrogen for long-term storage. For small batches of samples, a freezing box (filled with isopropanol) can be used to achieve a slow freezing rate (about 1℃/min).
- Pre-Cooling Treatment: Before freezing, the filled cryopreservation bags should be pre-cooled at 4℃ for 30-60 minutes to reduce the temperature difference between the sample and the freezing environment, avoiding thermal shock to the sample. Do not put the sample directly into ultra-low temperature environment for freezing, which will cause rapid formation of large ice crystals and damage the sample.
- Freezing Environment Requirements: The freezing operation should be carried out in a clean, sterile, and low-humidity environment to avoid condensation of water vapor on the surface of the bag body (which will cause the bag body to freeze and stick to the freezer, resulting in damage). After freezing, check the bag body for damage, leakage, or deformation, and discard the unqualified bags in time.
3.3 Long-Term Ultra-Low Temperature Storage
After the samples are completely frozen, they should be transferred to the ultra-low temperature storage environment in time for long-term storage. The key points of storage management are as follows:
- Storage Environment Selection: According to the sample storage requirements, select the appropriate storage environment. For short-term storage (1-6 months), a -80℃ ultra-low temperature refrigerator can be used; for long-term storage (more than 6 months), liquid nitrogen storage tanks (-196℃) are recommended, which can better maintain the activity of samples. The liquid nitrogen storage tank should be regularly checked for liquid nitrogen level, pressure, and temperature to ensure that the storage environment is stable.
- Storage Placement: The cryopreservation bags should be placed vertically or horizontally in the storage container, avoiding extrusion, collision, or sharp object scratching. The bags should be placed separately according to the sample type and batch to avoid cross-contamination and facilitate sample retrieval. For liquid nitrogen storage, the cryopreservation bags should be placed in a sealed storage box to avoid direct contact with liquid nitrogen (preventing the bag body from being damaged by liquid nitrogen immersion) and to reduce the volatilization of liquid nitrogen.
- Storage Period Management: Establish a complete sample storage management system, record the storage time, batch number, and retrieval situation of each sample, and regularly check the sample quality (such as cell viability detection) to ensure that the samples are not degraded. The storage period of samples should not exceed the expiration date of the cryopreservation bags, and expired bags should be replaced in time. For cell banks, regular subculture and preservation are recommended to maintain the stability of cell activity.
- Storage Environment Maintenance: The ultra-low temperature refrigerator and liquid nitrogen storage tank should be regularly maintained and cleaned, including defrosting, disinfection, and performance verification. The storage environment should be kept clean and dry, avoiding dust, moisture, and harmful gases, which will affect the performance of the cryopreservation bags and the quality of samples.
3.4 Sample Transportation
The transportation of biological samples is a key link to ensure sample quality between different institutions or departments. The core is to maintain the ultra-low temperature environment during transportation and avoid sample thawing or damage. The key points are as follows:
- Transport Container Selection: Use professional ultra-low temperature transport containers (such as liquid nitrogen transport tanks, dry ice transport boxes) to maintain the low-temperature environment during transportation. The transport container should have good thermal insulation performance, shock resistance, and sealing performance, and be able to maintain the specified temperature for a long time (usually more than 48 hours). For long-distance transportation, liquid nitrogen transport tanks are recommended; for short-distance transportation, dry ice transport boxes can be used (dry ice temperature is -78.5℃).
- Package Protection: Before transportation, the cryopreservation bags should be wrapped with shock-absorbing materials (such as foam, cotton) to avoid damage caused by collision and vibration during transportation. For critical samples, double-layer packaging (inner layer for sample storage, outer layer for shock absorption and insulation) is recommended, and a leakage-proof layer is added to prevent sample leakage in case of bag damage. In addition, the transport package should be marked with ""ultra-low temperature"", ""fragile"", ""biological sample"", and other warning signs.
- Temperature Monitoring: Place a temperature recorder in the transport container to monitor the temperature during transportation in real time, ensuring that the temperature does not exceed the specified range (for liquid nitrogen transportation, the temperature should be maintained at -196℃; for dry ice transportation, the temperature should be maintained below -70℃). After transportation, check the temperature record to confirm that the transportation process meets the requirements. If the temperature exceeds the specified range, the sample quality should be inspected in time, and unqualified samples should be discarded.
- Transportation Compliance: The transportation of biological samples must comply with the relevant regulations of global regulatory authorities (such as IATA, ADR) and local laws and regulations, especially for clinical samples and dangerous biological materials, which need to go through relevant approval procedures and be transported by qualified transport companies. The transport documents should be complete, including sample information, transport temperature requirements, and emergency handling measures.
3.5 Sample Thawing Process
The thawing process is the last link of the storage process, and the core is to control the thawing rate to avoid sample damage caused by ice crystal recrystallization. The key operation points are as follows:
- Thawing Rate Control: The optimal thawing rate for most biological samples is ""rapid thawing"", which can reduce the formation of ice crystals during thawing and avoid damage to the cell membrane. The common method is to place the cryopreservation bag in a 37℃ water bath (or thawing bath) and shake gently to make the sample thaw evenly. The thawing time should be controlled within 1-5 minutes (depending on the sample volume), and the sample should be taken out immediately after thawing (when there is a small amount of ice residue left) to avoid overheating and damage to the sample.
- Thawing Environment Requirements: The thawing operation should be carried out in a sterile environment (such as a biological safety cabinet) to avoid sample contamination. The water bath should be disinfected regularly, and the water should be replaced in time to avoid microbial growth. Do not thaw the sample at room temperature or in a high-temperature environment, which will cause rapid thawing and sample damage.
- Post-Thawing Treatment: After thawing, the sample should be processed in time according to the sample type. For cells, the cryoprotectant should be removed by centrifugation (usually 1000-1500 rpm, 5-10 minutes) to avoid the toxic effect of the cryoprotectant on the cells; for vaccines and biological reagents, the sample should be mixed evenly and placed on ice for use to maintain sample activity. After thawing, check the cell viability or sample quality, and discard the unqualified samples in time.
- Disposal of Used Bags: The cryopreservation bags after thawing and sample retrieval should be disposed of in accordance with the relevant regulations of biological waste, disinfected first (such as high-temperature sterilization, chemical disinfection), and then discarded to avoid environmental pollution and biological safety risks. Do not reuse disposable cryopreservation bags to avoid cross-contamination.
4. Key Principles of Cryopreservation Bag Selection and Risk Control
4.1 Key Selection Principles
The selection of cryopreservation bags should follow the principles of ""sample adaptation, safety first, and compliance with regulations"", and comprehensively consider the following factors to ensure that the selected cryopreservation bags can meet the actual application needs:
- Sample Type and Storage Requirements: According to the type of biological sample (cells, tissues, vaccines, blood products), storage temperature (-80℃ or -196℃), storage time (short-term or long-term), and sample volume, select the appropriate cryopreservation bag. For example, for stem cells and other high-value cells that require long-term liquid nitrogen storage, select multi-layer composite cryopreservation bags with good low-temperature resistance and barrier property; for small-volume experimental cells, select single-layer cryopreservation bags with small capacity.
- Compliance with Regulatory Requirements: The selected cryopreservation bags must comply with the requirements of global regulatory authorities (FDA, EMA, NMPA) and relevant international standards (ISO 11607, ISO 10993). The manufacturer should provide complete technical documents, including material certification, biocompatibility test reports, sterility test reports, and traceability records, to ensure that the cryopreservation bags can pass regulatory inspection and process verification.
- Performance Indicators: Prioritize the selection of cryopreservation bags with excellent performance indicators, including low-temperature resistance, biocompatibility, sealing performance, and mechanical strength. For critical samples, it is recommended to select products that have passed third-party verification to ensure product quality and stability. In addition, consider the compatibility with cryoprotectants and automated filling systems to improve application efficiency.
- Safety and Traceability: Select cryopreservation bags with closed sterile design to avoid sample contamination and leakage. The bag body should have a clear marking area and complete traceability information, and the manufacturer should provide a perfect after-sales service and technical support system to solve problems encountered in the application process. For clinical samples and cell therapy products, select cryopreservation bags with RFID tags to realize intelligent traceability and management.
- Cost-Efficiency: On the premise of meeting performance and safety requirements, comprehensively consider the cost of cryopreservation bags, including the unit price, storage cost, and transportation cost. For large-batch samples, select cryopreservation bags with large capacity to reduce the number of bags used and the overall cost; for non-critical samples, select cost-effective single-layer cryopreservation bags to balance cost and performance.
4.2 Key Risk Control Points
In the process of using cryopreservation bags, there are many potential risks (such as sample contamination, leakage, activity loss, and non-compliance), which need to be strictly controlled to ensure sample quality and process safety. The key risk control points are as follows:
- Contamination Risk Control: Strictly implement sterile operation in the whole process of sample preparation, filling, thawing, and retrieval, including the disinfection of the operating environment, instruments, and operators. Check the sterility of the cryopreservation bags before use, and discard the bags with damaged packaging or expired sterility. Avoid repeated opening of the bag body during sample retrieval to prevent microbial contamination.
- Leakage Risk Control: Check the integrity of the cryopreservation bag body and sealed port before filling, freezing, storage, and transportation. Ensure that the sealing operation is standardized, and the sealing strength meets the requirements. For critical samples, use double-layer packaging to further reduce the risk of leakage. Regularly check the bag body for damage during storage and transportation, and handle unqualified bags in time.
- Sample Activity Loss Risk Control: Strictly control the freezing and thawing rates to avoid sample damage caused by ice crystal formation and recrystallization. Select appropriate cryoprotectants and pretreatment methods according to the sample type to improve the cryopreservation effect. Maintain a stable ultra-low temperature environment during storage and transportation, and avoid sample thawing or overheating. Regularly check the sample quality to ensure that the sample activity meets the requirements.
- Compliance Risk Control: Establish a complete quality management system, including the verification of cryopreservation bags (performance verification, sterility verification), the standardization of operating procedures (SOP), and the traceability of samples. Ensure that the entire application process complies with the requirements of regulatory authorities and international standards, and keep complete operation records and verification reports for regulatory inspection. Regularly train operators to improve their professional skills and compliance awareness.
5. Technical Development Trends and Industry Challenges of Cryopreservation Bags
5.1 Technical Development Trends
With the continuous development of biopharmaceutical, clinical research, and cell therapy technologies, the technical level of cryopreservation bags is constantly improving, showing the following clear development trends:
- High-Performance Material Development: Developing new low-temperature resistant and biocompatible materials, such as modified EVA, polyimide (PI), and composite nanomaterials, to further improve the low-temperature resistance, mechanical strength, and barrier property of cryopreservation bags. At the same time, developing environmentally friendly and degradable materials to reduce the environmental pollution caused by disposable cryopreservation bags.
- Intelligent and Traceable Design: Integrating RFID, temperature sensor, and other intelligent components into cryopreservation bags to realize real-time monitoring of sample storage temperature, location, and state, and realize intelligent management and traceability of samples through the Internet of Things (IoT) technology. This can effectively avoid human error and improve the efficiency and safety of sample management.
- Customized and Specialized Development: According to the specific needs of different fields and sample types, developing customized cryopreservation bags. For example, for cell and gene therapy products, developing cryopreservation bags with small dead volume, high sealing performance, and traceability; for tissue and organ storage, developing cryopreservation bags with slow freezing and good biocompatibility; for vaccine storage, developing cryopreservation bags with good barrier property and compatibility with vaccines.
- Integration with Automated Processes: Optimizing the structural design of cryopreservation bags to improve their compatibility with automated filling, freezing, storage, and thawing systems, realizing the automation and intelligence of the entire sample storage process. This can reduce human error, improve work efficiency, and ensure the consistency of the process.
- Improvement of Cryopreservation Technology: Combining cryopreservation bags with new cryopreservation technologies (such as vitrification cryopreservation, ice crystal inhibitor addition), reducing the damage to samples during freezing and thawing, and improving the long-term storage effect of samples. For example, adding ice crystal inhibitors to "