1. Introduction
In modern biological and medical fields, the long-term preservation of biological samples—including cells, tissues, stem cells, CAR-T cells, hematopoietic progenitor cells, and vaccines—is critical for clinical treatment, scientific research, and biopharmaceutical production. Unlike traditional rigid cryovials, cryopreservation bags offer larger storage capacity, better flexibility, and improved compatibility with ultra-low temperature environments, addressing the limitations of vials in large-volume sample storage and transportation. They are designed to withstand extreme temperature fluctuations, prevent sample damage from ice crystal formation, and maintain sterility throughout the cryopreservation process. With the rapid development of cell therapy, regenerative medicine, and biobanking, the global cryopreservation bag market is growing steadily, with a projected CAGR of 4.0% from 2025 to 2031, driven by increasing demand for high-viability sample preservation and stricter GMP compliance requirements.
2. Structure and Working Principle
2.1 Core Components
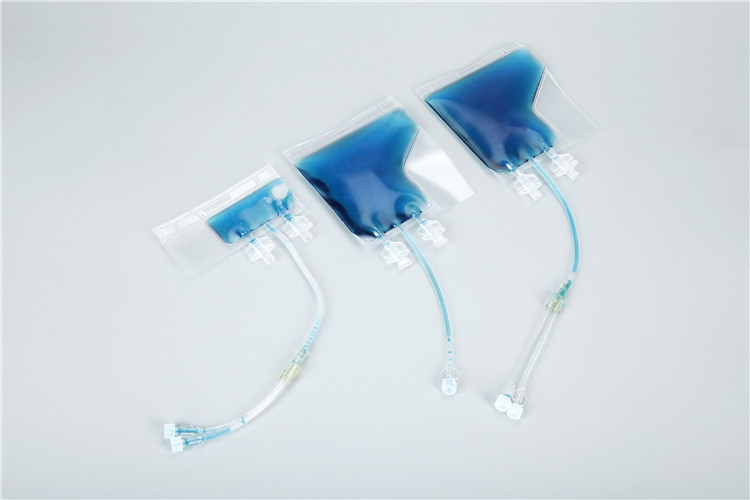
- Bag Body: The main container, typically made of a proprietary blend of medical-grade polyolefin and EVA (ethylene-vinyl acetate) or multi-layer co-extruded films (e.g., PET/PE/EVOH), selected for their excellent low-temperature flexibility, biocompatibility, and resistance to liquid nitrogen infiltration. The film remains flexible even at -196°C, preventing cracking or rupture during freezing and thawing. Some models feature a transparent design for easy visual inspection of sample volume and state, while others are available in opaque versions for light-sensitive samples.
- Sealing System: A critical component featuring heat-sealable edges and specialized ports with attached caps. The heat-sealing design ensures a hermetic, leak-proof seal to maintain sterility and prevent sample contamination, while the attached caps minimize membrane exposure during freezing and handling. The ports (usually 1-2 per bag) are designed with thin-walled membranes for easy access and compatibility with sterile connection technology and tube welders, facilitating safe sample transfer without breaking the sterile barrier.
- Volume Markings: Clear, calibrated graduations (in milliliters) on the bag body to ensure accurate sample loading, typically ranging from 10 mL to 750 mL. The recommended fill volume is usually 80% of the bag’s total capacity to accommodate liquid expansion during freezing, reducing the risk of rupture.
- Labeling Area/Pocket: A dedicated, cryo-resistant surface or built-in label pocket for attaching computer-generated or handwritten labels, enabling easy sample identification and traceability. This design supports compliance with GMP requirements by ensuring clear documentation of sample information (e.g., sample type, donor ID, collection date, freezing protocol).
- Optional Features: Some cryopreservation bags include multi-chamber designs (2-5 independent compartments) for parallel storage of multiple samples, reducing cross-contamination and sample waste. Others may be pre-sterilized (gamma-sterilized) and packaged in double-layer packaging to ensure sterility before use.
2.2 Working Principle
Cryopreservation bags operate based on the principle of controlled ultra-low temperature storage, working in conjunction with cryoprotectants (e.g., DMSO, glycerol) to minimize ice crystal formation and protect biological samples from damage. The flexible, low-temperature-resistant bag body accommodates liquid expansion during freezing (up to 9% volume expansion) without rupturing, while the hermetic seal prevents contamination and liquid nitrogen infiltration. During freezing, the bag is typically placed horizontally in a protective cassette to ensure uniform cooling, with a recommended internal thickness of approximately 8 mm for optimal freezing efficiency. The controlled cooling rate (usually -1°C per minute) minimizes ice crystal formation, which can damage cell membranes and reduce sample viability. During thawing, the flexible bag allows for rapid, uniform warming (e.g., in a 37°C water bath), further preserving sample integrity. The entire process maintains the sample’s biological activity, enabling long-term storage (months to years) at -80°C or -196°C.
3. Classification and Key Features
3.1 Classification
- By Material: Polyolefin-EVA blend bags (most common, offering excellent flexibility and biocompatibility at ultra-low temperatures) and multi-layer co-extruded bags (e.g., PET/PE/EVOH, providing enhanced barrier properties to prevent gas permeation). All materials meet USP Class VI or ISO 10993 biocompatibility standards to ensure no adverse reactions with biological samples.
- By Chamber Design: Single-chamber bags (for large-volume, homogeneous sample storage, such as bulk stem cell or cell line storage) and multi-chamber bags (for parallel storage of multiple small-volume samples, ideal for personalized medicine, and experimental research).
- By Capacity: Small-capacity (1-25 mL, for rare samples like CAR-T cells and iPSCs), medium-capacity (50-250 mL, for routine cell lines and hematopoietic progenitor cells), and large-capacity (500-750 mL, for industrial-scale biopharmaceutical production).
- By Application: Cell therapy bags (for CAR-T cells, mesenchymal stem cells), hematology bags (for hematopoietic progenitor cells and), biopharmaceutical bags (for vaccine and cell line storage), and auxiliary bags (for oocytes, sperm, and embryos).
3.2 Core Features
- Ultra-Low Temperature Resistance: Remains flexible and intact at temperatures as low as -196°C (liquid nitrogen), preventing cracking or rupture during freezing, storage, and thawing.
- Biocompatibility: Made of USP Class VI or ISO 10993 certified materials, ensuring no cytotoxicity, no sample adsorption, and no adverse reactions with biological samples, preserving cell viability and functionality.
- Hermetic and Leak-Proof: Heat-sealable edges and specialized port designs create a secure, sterile seal, preventing contamination, liquid nitrogen infiltration, and sample leakage.
- Flexible and User-Friendly: Lightweight, flexible design facilitates easy handling, storage, and transportation, while clear volume markings and label pockets simplify sample identification and traceability.
- Sterility: Most models are pre-sterilized (gamma-sterilized) and supplied in double-layer packaging, ensuring sterility for clinical and biopharmaceutical applications.
4. Proper Usage and Maintenance
4.1 Correct Usage Steps
- Select the appropriate cryopreservation bag based on sample type, volume, and application. For sensitive samples (e.g., oocytes), choose multi-chamber small-capacity bags; for bulk cell storage, select single-chamber large-capacity bags.
- Inspect the bag for damage (e.g., tears, pinholes, or defective seals) before use; discard any damaged bags to prevent leakage or contamination. Ensure the bag is within its expiration date and properly sterilized.
- Prepare the sample by mixing it with an appropriate cryoprotectant (e.g., 10% DMSO) to minimize ice crystal damage. Load the sample into the bag, filling it to no more than 80% of the total capacity to accommodate liquid expansion during freezing.
- Expel all air from the bag before sealing to reduce ice crystal formation and ensure a secure seal. Use a validated heat sealer to seal the bag edges, applying even pressure and appropriate temperature/time settings based on the bag material. Inspect the seal post-processing for leaks.
- Label the bag immediately with all necessary information (sample ID, donor information, collection date, cryoprotectant type, and freezing protocol) using a cryo-resistant label.
- Freeze the bag using a controlled-rate freezer (for GMP facilities and commercial products) or an IPA-free cooling box (for laboratory research), ensuring a cooling rate of approximately -1°C per minute. Do not place the bag directly into liquid nitrogen without gradual cooling.
- Store the frozen bag in liquid nitrogen (-196°C) or a -80°C freezer for long-term preservation. For transportation, use a cryogenic container to maintain ultra-low temperatures and prevent thawing.
- Thaw the bag by submerging it in a 37°C water bath (≤40°C) for 2-3 minutes until the ice melts, gently agitating to ensure uniform warming. Avoid contact between the bag and water if sterility is critical, using a sealed secondary bag if necessary. Inspect the bag for leaks before using the sample.
4.2 Maintenance and Hygiene
- Disposable Bags: Most cryopreservation bags are single-use (intended for one-time use) to prevent cross-contamination. Discard the bag after thawing and sample retrieval; do not reuse, as the seal may be compromised and residues may remain.
- Storage of Unused Bags: Store unused cryopreservation bags in a clean, dry, dust-free environment, away from direct sunlight, extreme temperatures, and corrosive substances. Keep them in their original packaging until use to maintain sterility.
- Sealer Maintenance: Regularly calibrate and maintain heat sealers to ensure consistent, secure seals. Clean the sealer surface to remove any residue that may interfere with sealing efficiency.
- Traceability: Maintain detailed records of each bag’s usage, including sample information, storage conditions, and thawing procedures, to comply with GMP requirements and ensure sample traceability.
5. Applications Across Industries
Cryopreservation bags are widely used across biological, medical, and biopharmaceutical industries, each with specialized requirements:
- Cell Therapy: The core application field, used for the storage and transportation of CAR-T cells, mesenchymal stem cells, and other therapeutic cells. They ensure high cell viability post-thaw, supporting the clinical application of cell-based therapies for cancer and other diseases.
- Hematology and Stem Cell Transplantation: Used for the preservation of hematopoietic progenitor cells, and bone marrow cells. They are essential for banks, leukemia treatment, and bone marrow transplant procedures, maintaining stem cell proliferation and differentiation capacity.
- Biopharmaceuticals: Used for the storage of cell lines (e.g., CHO cells for monoclonal antibody production) and vaccines during research, development, and production. They meet GMP requirements, ensuring sample integrity and traceability.
- Auxiliary Reproduction: Used for the preservation of oocytes, sperm, and embryos, supporting fertility preservation and infertility treatment. Multi-chamber bags are often used to store multiple samples from the same donor, reducing waste.
- Laboratory Research: Used for the storage of cell lines, tissues, and biological samples in academic and research institutions, facilitating long-term experimental studies and sample sharing.
6. Common Issues and Troubleshooting
- Bag Rupture: Caused by overfilling (exceeding 80% capacity), rapid temperature changes, or damaged bags. Solution: Adhere to the recommended fill volume, use controlled-rate freezing and thawing, and inspect bags for damage before use.
- Leakage: Resulting from improper heat sealing, damaged ports, or worn caps. Solution: Ensure proper heat sealer calibration, inspect seals post-processing, and replace damaged ports or caps. Perform a leak test by submerging the sealed, empty bag in water and applying gentle pressure.
- Low Sample Viability: Caused by inadequate cryoprotectant use, rapid freezing/thawing, or incompatible bag materials. Solution: Use the appropriate cryoprotectant concentration, follow controlled cooling and thawing protocols, and select biocompatible bags.
- Contamination: Caused by non-sterile bags, improper handling, or seal failure. Solution: Use pre-sterilized bags, maintain sterile technique during sample loading and sealing, and discard any contaminated bags.
- Poor Traceability: Caused by incomplete labeling or inadequate record-keeping. Solution: Label bags immediately with all required information and maintain detailed records of storage and handling procedures.
7. Development Trends
The global cryopreservation bag market is evolving to meet the growing demand for precision, safety, and efficiency in biological sample preservation. Key trends include the development of specialized materials with enhanced barrier properties and lower cytotoxicity, tailored to specific sample types. Smart technologies are being integrated, such as RFID tags for real-time temperature monitoring and sample tracking, improving traceability and compliance with GMP requirements. Additionally, multi-chamber designs and sterile connection compatibility are becoming more prevalent, facilitating personalized medicine and automated bioprocessing. There is also a growing focus on sustainability, with the development of eco-friendly, biodegradable materials to reduce medical waste. Furthermore, advancements in cryopreservation technology are driving the design of bags optimized for these techniques, further improving sample viability.
8. Conclusion
Cryopreservation bags are indispensable specialized tools for the long-term preservation of biological samples in biopharmaceuticals, cell therapy, clinical medicine, and scientific research. Their ultra-low temperature resistance, biocompatibility, hermetic sealing, and user-friendly design ensure that biological samples maintain viability and integrity during freezing, storage, and transportation. As the global demand for cell-based therapies, biobanking, and biopharmaceutical production continues to grow, cryopreservation bags will play an increasingly critical role in supporting advancements in precision medicine and life sciences. Ongoing innovations in material science, design, and smart technology will further enhance their functionality, making them more efficient, safe, and adaptable to the evolving needs of the biological and medical industries.